“Investigator sites that don’t understand new privacy rules that went into effect this year in many states will find it very difficult to conduct clinical trials in 2021,” says Al […]
The U.S. Food and Drug Administration (FDA) recently released a final guidance detailing the process for sponsors and clinical researchers to submit plans for new drug development tools (DDTs) […]
CISCRP Campaign, Other Initiatives Aim to Promote Patient and Workforce Diversity in Clinical Trials
The topics of promoting patient diversity in the clinical trial population and workforce diversity in the profession continue to receive increased and welcome focus from the drug and device research […]
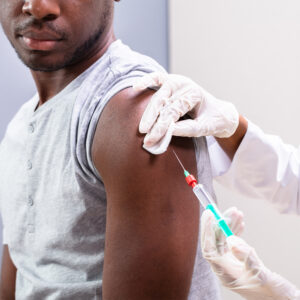
While the clinical trial industry has performed incredibly well in terms of developing potential vaccines against COVID-19, the next big challenge is to allay concerns many have about taking any […]
The U.S. Food and Drug Administration (FDA) “encourages diverse patients to participate in medical device clinical trials,” the agency says in part of a new series of videos released […]