Clinical Researcher—March 2019 (Volume 33, Issue 3)
GOOD MANAGEMENT PRACTICE
David J. Morin, MD, FACP, CPI, FACRP
Accountability is the process by which an individual or organization is held responsible for a set of accepted rules. Sites should develop thorough accountability processes for the protection of subject welfare and data as a good management practice internally, and because they are held accountable by external regulatory agencies. There is also a chain of accountability connecting site staff to their manager, the manager to the organization, and the organization to governing agencies. For such an important process, however, there is very little information available on how to create it.
Take a minute and ask if your research site has methods in place which genuinely allow individuals to be held accountable for their actions. For example, when addressing an issue, have you ever heard, “That’s not how I was taught to do it” or “I didn’t know that was my responsibility”? However, what if their training failed to address a task or they were not assigned the authority to perform a particular function?
This brief article will detail the steps required to create accountability at the research site level. Such accountability allows sites to hold individuals responsible for their assigned roles, but also to bring managers to account for gaps in the process and for updating the system when required.
The Essential Steps

Accountability begins by assigning responsibility in your standard operating procedures (SOPs). Though SOPs are not regulatory requirements, they are essential to creating a uniform standard of behavior across the spectrum of research site actions. They follow the regulations and allow the site manager to assign tasks to individuals based on their study roles as a result of their level of education, training, and experience. They define expectations and delegate authority inherent in the company role. SOPs can be acquired commercially and then modified to your site requirements or thoroughly developed internally. However, if you have SOPs, you are required to follow them.
Once each role is appropriately assigned, this information is listed directly from the SOP to create a job description. The job description is separately added to the SOP and sent to prospective employees before a first interview. By sending this information at the pre-employment phase, you are setting expectations very early. I added this process where I work years ago, after hiring a promising new clinical research coordinator who was shocked to learn on her first day that she was expected to have contact with people! We never said the position was purely lab based, but hadn’t provided a list of job expectations. Because this wasn’t clearly written down, wrong assumptions were made on both sides. Providing the list of job duties very early in the process fixed that issue. However, it also had the possibility of discouraging novice applicants, so we reassure a promising candidate that we will train them to perform all assigned tasks.
As soon as a prospective candidate is hired, begin the training process to include all tasks assigned in their job expectations. Also, include ancillary items required of all employees. At our institution, we use a training program developed internally and appoint an experienced mentor. To preclude boredom and keep the training relevant to the roles, it is a mix of didactic and clinical education. Also, training programs may be obtained commercially.
Mentoring allows supervision and oversight to help verify competency, which is the ability to perform one’s job effectively. We utilize a training checklist and regularly meet with the new employee to assess progress and skill. Each training section is divided into separate areas which are initialed and dated by the mentor and manager once completed. Finally, we keep this training record on permanent file should we need to verify training at some future date.
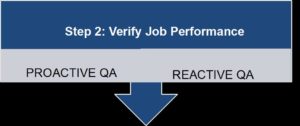
Verifying job performance is an ongoing process which is used to assure regulatory compliance and employee competency. Examples of a “proactive” quality assurance (QA) process are the secondary review of all source documents before screening subjects, and of subjects before randomization. This process is used to help prevent protocol deviations. Reactive or “retroactive” QA processes review completed work; they may respond to an internal concern or an issue discovered by the study monitor, and may be done in preparation for an external audit or on a routine basis. Competency is confirmed when the quality of the work is acceptable.
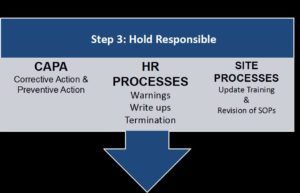
Finding an activity which falls outside the scope of acceptable practice, or outside the boundaries as defined by the protocol, requires immediate attention. There are many good articles on “Corrective Action and Preventive Action” (CAPA) for readers to review. Whether the improper action requires a CAPA or process involving the Human Resources (HR) department is determined by the manager, based on the severity and intent of the action.
CAPAs are used to address a process that is inconsistent with the expected performance tasks. They are part of an administrative process used to show “due diligence.” It’s possible that the action occurred as a result of inadequate training. In this case, the new process is added to the SOP along with documented training for all affected employees.
While CAPAs are educational, a “Warning” or “Write Up” is more disciplinary in nature. We strictly follow the recommendations of our HR department when disciplinary actions occur.
Figure 1 depicts how I see this process as continual. New regulations are entered into the SOP and assigned to those responsible. The defined expectations for a given job position are used as the basis for training. The next section along the “wheel” is to assess job performance and verify competency through QA programs. The final part is to address and correct any deficiencies by site actions, which include CAPA for processes requiring additional training and due diligence to actions which are more punitive. Any methods which are new or updated add to the SOP (at the top), and the process begins again.
Figure 1: Closing the Circle of Accountability
A recent example of changes to our SOP occurred when we received a study which randomized the subject the same day of screening. We allowed some flexibility to review the first two subjects within 24 hours after randomization if it could not be done earlier. By following these general processes, all levels of the organization are held accountable to their roles and expectations, including management.
Conclusion
Developing a thorough accountability process may take time, but it is not complicated. It permits modification at any time based on the site needs. Moreover, the time invested will reap big rewards by creating an environment of stability.
David J. Morin, MD, FACP, CPI, FACRP, is Director of Research with The Holston Medical Group in Kingsport, Tenn., and a member of the Association Board of Trustees for ACRP.



