Clinical Researcher—January 2020 (Volume 34, Issue 1)
RECRUITMENT & RETENTION
Neetu Pundir; Mika Lindroos; Jenna McDonnell; Bill Byrom; Spencer Egan
With the objective of learning more about the industry’s global experiences and perspectives with electronic informed consent (eConsent), Signant Health conducted a survey in 2019 that sought to obtain insights from more than 130 respondents working in sponsor settings and with contract research organizations (CROs). This column presents findings from this key piece of industry research.
Background
For several years now, eConsent has been making a name for itself as a technological gamechanger in clinical trials. With industry groups, academic research centers, and, crucially, regulators advocating its innovative advantages, it is no surprise that many of the major pharmaceutical companies are keen to explore eConsent initiatives.
In 2017, CRF Health (now Signant Health) conducted its first survey to dig a little deeper into the industry’s attitudes toward eConsent. This enabled industry experts to understand and to benchmark its current use, better understand adoption hurdles and challenges, and anticipate future use and adoption. Two years on, it is interesting to take a fresh look at how those in the industry view eConsent, and to learn from the experiences of companies employing the technology—from extensive use to pilot testing.
The 2019 Industry eConsent Survey received responses from personnel at sponsors and CROs who have had hands-on experience with implementing eConsent or were working with teams directly involved in its planning and implementation. The survey was not specific to any eConsent solution or provider.
The profiles of respondents covered a wide range of therapeutic area responsibilities as well as functional groups, including executives, clinical operations, regulatory, data management, innovation, information technology, clinical sciences, and procurement. Some of the key areas from the survey presented and discussed in this article include uptake rates by study phase, experiences, and feedback on operational services provided by eConsent solution providers, and which aspects of eConsent solutions were considered to have a high impact on patients’ comprehension and engagement.
The results also demonstrate increasing demand to integrate consent data with other eClinical systems (e.g., electronic data capture [EDC] systems) and key market trends and perspectives on eConsent deployment in clinical trials by early adopters. Other highlights include areas of improvement, level of satisfaction, and remaining challenges for eConsent solutions and implementation. Finally, the report provides some clarity on the business drivers behind decisions about whether to implement eConsent.
The Respondents
The survey captured the opinions of 134 respondents from predominantly biotech and pharmaceutical companies (60%) and CROs (34%). Thirty-nine percent of respondents worked in clinical operations functions, with a further 13%, 8%, 7%, and 7% in health outcomes, procurement, data management, and regulatory functions, respectively. Therapeutic area responsibilities covered a broad range including oncology, central nervous system, immunology, gastrointestinal, pediatrics, hematology, endocrinology, respiratory, and dermatology, amongst the most common.
eConsent Experience and Adoption
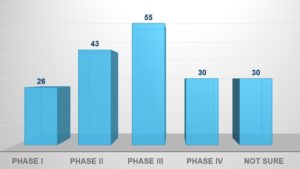
Respondents varied in their levels of experience with implementing eConsent solutions (see Figure 1). Thirty-one percent had implemented an eConsent solution in anywhere from one to five studies, 18% in six to 15 studies, and a further 18% had implemented more than 15 studies (remainder not answered or unsure). Fifty-five respondents (41%) indicated usage in Phase III trials, with 19%, 32%, and 22% in Phases I, II, and IV, respectively (see Figure 2).
From those asked, North America was the most popular region where eConsent has successfully been implemented (72%), followed by Europe (37%).
Figure 1: Profile of Respondents—How Much Experience Does Your Business Unit Have with eConsent?
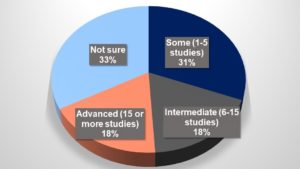
Figure 2: For Which Phase is eConsent Typically Used at Your Organization?
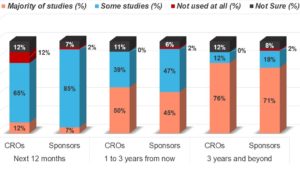
Indeed, the momentum of adoption looks set to continue with 65% of CRO respondents and 85% of sponsor respondents saying that their organizations will adopt eConsent for some studies over the next 12 months. While in the long term, 76% of CRO respondents and 71% of sponsor respondents state that their organizations will adopt eConsent for the majority of studies in the next three years and beyond (see Figure 3).
Figure 3: The Future of eConsent—How Do You Predict eConsent Use at Your Organization?
Business Drivers and Impact of eConsent
Respondents were asked to rank the importance of a number of potential business drivers for implementing eConsent in their organizations. Most commonly identified in the top three business drivers were improved patient comprehension, efficiencies through digitization, improved patient retention, and reduced regulatory risk and audit findings. The top drivers identified were similar between biopharmaceutical company respondents and CROs.
Related to these top business drivers, further exploration of aspects of eConsent that may impact patient comprehension and engagement found 91% of CRO respondents and 73% of sponsor respondents reported a user-friendly and interactive interface (e.g., glossary of complex terms) was a highly impactful feature. Other features of eConsent thought to highly
impact patient comprehension and engagement were the ability for patients to flag questions to discuss with site staff (73% and 69%), multimedia tools (91% and 50%), and the ability to read consent documents at home (64% and 56%) (figures are CRO and sponsor respondents, respectively).
This is in line with what the U.S. Office for Human Research Protections (OHRP) and the U.S. Food and Drug Administration (FDA) stated in a December 2016 guidance document on the use of eConsent in clinical trials. It was noted that “electronic processes to obtain informed consent may use an interactive interface, which may facilitate the subject’s ability to retain and comprehend the information.”
Satisfaction and Challenges
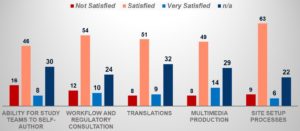
From an eConsent design and development perspective, 63% of respondents reported that they were satisfied with the setup processes such as procuring digital tablets for site use, training sites, and creating site users. However, 16% were not satisfied with the ability for study teams to easily configure and self-author eConsent without vendor involvement (see Figure 4).
The reliance on outside vendors to design and program eConsent can slow down implementation and create longer timelines in making the eConsent “live.” Consequently, sponsors and CROs are looking for adaptable solutions and support models that enable them to control and manage the design, development, and deployment of eConsent without the need for continuous vendor support. While the majority of respondents were largely satisfied, there is room for improvement to get to “very satisfied.”
Figure 4: eConsent Experience—Rate Your Organization’s Level of Satisfaction on Items Related to Design and Development of eConsent Forms
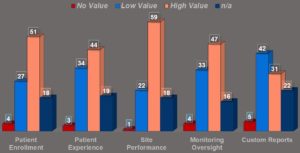
From a data reporting perspective, 59% of respondents placed high value on site performance reporting, such as enrollment rate, pending re-consent. This was closely followed by patient enrollment data (51%), monitoring oversight (47%), and patient experience (44%). Most data
are considered highly valuable, while custom reports were identified as relatively low in value (see Figure 5).
Figure 5: What Reporting Data from eConsent Systems Have Been Most Valuable to You?
One of the biggest challenges reported with using eConsent in clinical trials was investment in new technology. As with all new technology, perceived high costs and uncertain return on investment can prove to be a barrier and prevent some organizations from adopting eConsent, with 38% of respondents reporting that investment in new technology can be challenging.
Often, the costs of existing processes associated with consent form development, implementation, and monitoring are not measured, nor the costs of loss of data due to ambiguous or incomplete informed consent documentation or resulting regulatory findings. While site support was identified as a potential challenge by 37% of respondents, those with more experience (>15 studies using eConsent) reported this less often—with only 15% of these advanced users citing this as a challenge (see Figure 6). This demonstrates the learning curve during the adoption phase, common to the implementation of all new processes or technologies.
Figure 6: What Have Been the Biggest Challenges with Using eConsent for Your Studies?

The Importance of Data Integration
Perhaps giving a more revealing glimpse into the industry’s current mindset were the responses around eClinical systems integration. Twenty-eight percent of respondents had integrated an eConsent solution with an EDC product, with the same number (28%) reporting integration with a clinical trial management system. A further 26%, 19%, and 17% had integrated eConsent with electronic clinical outcome assessment, electronic trial master file, and randomization and trial supply management solutions. Integration experience was largely positive.
Surprisingly, 36% of respondents had not integrated eConsent with other eClinical systems, and this may be a feature of the early adoption phase for this technology as the importance and drive to simplify processes and limit reconciliation through eClinical integration continues to be a focus within our industry.
Conclusion
Reflecting on the findings of the survey, eConsent is a valuable technology for many organizations in the drive to improve clinical trial processes, efficiency, and quality, and to generate a better patient experience impacting patients’ comprehension and ongoing engagement. This is further underlined by the intent of many companies to significantly ramp up their use of eConsent solutions in the next three years, as illustrated by our survey findings.
Understanding the features of solutions that most impact the improvement of patient comprehension and patient and site experience helps to focus ongoing development of these solutions. It is encouraging to see the continued and increased interest in eConsent now being realized with increased utilization within biopharmaceutical companies and CROs—a trend that is growing rapidly.
With increased adoption, companies can expect to benefit from internal efficiencies in addition to the secure knowledge of well-informed and engaged patients; transparency, clarity, and regulatory adherence around the consenting event; and simplified monitoring oversight in terms of consent tracking, document version control, and revision implementation remotely.
The benefits reflected clearly by responses to this survey act as a pulse check on attitudes and intent around eConsent. As a result, as an industry we will soon transition from the early adopter stage to mainstream use of this technology.
Neetu Pundir is Director of Go to Market Strategy, Product Management for Signant Health.
Mika Lindroos is Director of Project Management, eConsent for Signant Health.
Jenna McDonnell is a Program Manager for Signant Health.
Bill Byrom is Vice President for Product Strategy and Innovation for Signant Health.
Spencer Egan is Senior Director of Business Development for Signant Health.



