Clinical Researcher—April 2026 (Volume 40, Issue 2)
SPECIAL FEATURE
Jessica Fritter, DHSc, MACPR, ACRP-CP, FACRP; Susan P. Landis; Samantha Sharpe, MD; Tia Patterson, PhD; Kimberly McCall, PhD; Samantha Martinek Bundt, MA; Tiffany Stoudemire, MCR; Justin Osborne, BA; Carolynn T. Jones, DNP, MSPH, CRN-BC, FAAN
Clinical research professionals are members of a profession that has grown exponentially and evolved rapidly. Clinical research as a distinct profession has followed the path of many other groups of expert workers, with training moving from informal education (e.g., “see one, do one”) to formal certification along with recognition by academic, industry, and government entities.{1–3}
While recognized as an established and respected profession by academic medical centers (AMCs), pharmaceutical sponsors, and contract research organizations (CROs), we think the public is unaware of the profession and believe there are even those in the profession who have little to no knowledge about the breadth of career possibilities in the industry. We are raising greater awareness by actively peeling back the layers on myriad lucrative career pathways available in clinical research.
Well, How Did We Get Here?
Generally, clinical research professionals have found themselves in this ecosystem “by accident”—often saying that they unwittingly “fell into it” without clear and deliberate career entry points and progression pathways, and with some transitioning from other health-related fields and others from non-health sectors.{4,5} The ultimate goal is for future professionals in this enterprise to proactively jump into it rather than fall in.
In addition to lack of awareness of this career pathway, there can be a lack of consistency across requirements to enter the profession, including specific requirements for: experience, education, and certification. Often human resource departments are equally in the dark about these professions, and only recently have been included in strategic discussions to remedy this gap.{6,7}
While there still can be barriers to entering the clinical research profession, these are being reduced by intentional approaches to expand the clinical research workforce. There has been considerable advancement in deliberate entry points into the profession’s roles through educational efforts (e.g., academic programs, internships, and apprenticeships), building professional awareness (outreach to middle/high schools and junior colleges), and building more uniform job descriptions that offer logical progression.{8}
The adoption of the Joint Task Force Competency for Clinical Trials as an industry standard has been an inflection point within the profession, providing a framework of a cohesive and standardized set of knowledge, skills, and abilities within defined competency domains.{9,10} This has contributed directly to the realignment of job descriptions, educational curricula, career progression, and career pathway development.
How Do We Know Where We’re Going?
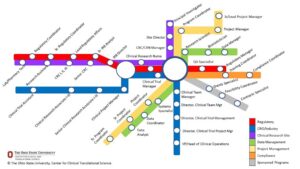
A visualization of the vast majority of career options has been illustrated via the “Train Tube Stops Model,” published by Fritter and Jones in 2023 and seen in Figure 1.{11} This has been utilized in an initiative by the Association of Clinical Research Professionals (ACRP) to petition the U.S. Bureau of Labor Statistics to assign an official code for these roles as a coherent profession.{4} However, as robust a start as it was for illustrating career pathways and for prompting discussions about the facilitators and barriers to entering the profession, it was not an all-inclusive model for the enterprise.
We have taken a broader look at our profession to be more inclusive of the career opportunities found with pharmaceutical sponsors, CROs, commercial institutional review boards (IRBs), and technology solution vendors, so we have redirected our vision from the “tube stop” concept to the “Airport Concourse Model” described next.
Figure 1: A Model of Clinical Research Professional Careers as Train Tube Stops
To the Airport!
Our new Airport Concourse Model (see Figure 2) provides an illustration for future and current professionals to navigate this enterprise with seamless intention and to not feel as if they are in a mad dash in an airport, seeking the right gate in a hurry, like some of the “accidental” career journeys of which we have heard.
A working group was formed to ensure representation of individuals working across different sectors in clinical research, including AMCs, clinical research sites, pharmaceutical sponsors, CROs, and commercial IRB. The group met monthly to brainstorm on current roles and job ladders, leading to a transformation of the “tube stop” illustration into a larger hub airport. We also invited external reviewers who worked in these areas to ensure we had not left off key roles. Additionally, we interviewed individuals working across sectors for input so that we could create vignettes representing real-life career stories across roles.
Figure 2: A Model of Clinical Research Professional Careers as an Airport Concourse

Job descriptions for all roles found in this model are explained in a downloadable PDF: Airport Concourse Model_Final.
Traversing Across the Concourses
The “concourse train” under the “career airport” is the way individuals may travel forward in their careers from one concourse to the other, or within a concourse. Factors that influence progression from one job category or within a job category include: education, experience, specialized training, certifications, individualized development planning, mentoring, passion drivers, and scholarship.
Our airport represents a generalized approach to illustrating job categories, and we will illustrate our findings across each stakeholder (employer) area.
Concourse A: Pharma Drugs
When clinical research professionals who are working as study coordinators or managers are asked to identify key roles within pharmaceutical sponsors, many initially think of the clinical research associate (CRA) or study monitor responsible for site selection, site initiation, and ongoing monitoring activities. This perception is understandable because the CRA is typically the sponsor or CRO representative with whom site staff interact most frequently and is their primary point of contact with these project partners.
However, the scope of roles within sponsor organizations extends far beyond traditional monitoring functions. Workforce shortages and increasing trial complexity have prompted greater visibility into the diverse career pathways available within pharmaceutical sponsors and CROs. ACRP initially described some of these roles in its 2020 Career Lattice.{12} Since then, new resources have provided some web-based insight into these roles.{13–15} These resources have further expanded awareness of the breadth of opportunities across the industry.
Within pharmaceutical sponsor organizations, roles typically fall into multiple functional groupings, often described broadly across eight core categories, each encompassing numerous position titles and levels of responsibility (see Figure 3).
In addition to clinical monitoring, sponsors maintain dedicated teams responsible for study start-up, regulatory submissions, contract and budget negotiations, and site activation to support outsourced trials. Sponsor-based site engagement and global site management roles focus on cultivating investigator relationships, optimizing site performance, and ensuring alignment between sponsor strategy and CRO-executed operations. These functions are particularly essential in fully or partially outsourced models, where sponsors retain strategic oversight, performance management, and accountability for trial delivery while delegating operational execution to CRO partners.
Collectively, these roles illustrate the multifaceted infrastructure required to support modern clinical development programs and demonstrate that sponsor organizations offer diverse career pathways beyond traditional monitoring positions.
Figure 3: Categories of Clinical Research Positions in a Pharmaceutical Sponsor

Concourse B: Pharma-Biotech/Biologics
In addition to synthetically made medical treatment products, cellular and biological materials are also used to create new medicinal products; this space is referred to as biotechnology (biotech). Although biotechnology companies as a whole span across multiple sectors such as environmental, industrial, farming, and healthcare, products generated for the purpose of providing medical treatment to humans must go through the clinical trial process to obtain regulatory approval before it can be marketed publicly.{16}
Biotech companies may utilize CROs to fulfill some or all the functions needed to operate their clinical trials and career pathways. In Concourse D, you will learn more about that sector, but you will find there is some overlap in structure between CROs and biotech companies.
Concourse C: Pharma-Devices/Assays
According to the U.S. Food and Drug Administration (FDA), medical devices can be as simple as a tongue depressor or as complex as a pacemaker for the human heart. Devices are tools that are used to diagnose, treat, cure, prevent, or mitigate diseases, trauma, or other medical issues through non-biological means.{17} Devices that pose a risk must go through a clinical trial process to determine safety and efficacy.
Medical devices may sometimes be used with a drug or biologic in tandem, and in such cases (like evaluating a new diabetes medication that is within a new type of injection pen) both the treatment and mechanism are part of the trial. The career paths and job titles in this sector closely mirror those within the pharma and biotech companies.
Concourse D: Contract Research Organization
Entry into CROs rarely follows a single, predefined professional path. Many individuals move into CRO roles after gaining experience in academic research settings, clinical trial sites, or broader healthcare environments. Prior involvement in study coordination, clinical data oversight, and trial implementation often provides essential preparation in regulatory processes, protocol adherence, and operational management that translates effectively to sponsor-delegated responsibilities within CROs.
As pharmaceutical and biotechnology sponsors have expanded the outsourcing of clinical development functions to enhance efficiency and their global footprint, CROs have become a common career destination for research professionals seeking participation in large, multicenter, and global trials, clinical monitoring activities, and collaborative operational teams.{18} Career transitions into CROs are frequently facilitated by professional networking, continued training, and exposure to industry-sponsored studies.
In response, many CROs have developed formal onboarding and training initiatives to help site-based research staff successfully adapt to industry-oriented roles. Further, CROs have diversified into more specialty areas (i.e., data management, study management, and supply chain). We expect to see CROs continue to expand as the clinical research industry grows.
Concourse E: Academic Medical Center/Site
AMCs employ a vast number of clinical research professionals to conduct numerous clinical studies, with the professionals serving as the operational backbone, ensuring timely start-up, regulatory compliance, and quality across the trial lifecycle. Newcomers often struggle to see a clear entry path; workforce analyses consistently cite role ambiguity, uneven onboarding, and inconsistent progression structures as barriers for new and current professionals at AMCs.
Navigating inconsistent job titles, limited career visibility, and varying expectations for experience are also challenges in this setting.{4–6} These realities make it difficult for new professionals to interpret positions and practical opportunities for career growth, especially considering how trial-related roles are both clinical and non-clinical positions.
Even when consistent job titles with corresponding ladders and competencies exist in theory at an AMC, such issues as inconsistent processes and vague criteria for applying them in practice, as well as limited funding capabilities for promotions, can negatively impact advancement opportunities. However, clinical research professionals continue to be integral to the success of clinical trials at AMCs, as the research conducted showcases true translational science from bench to bedside.
Concourse F: Commercial Institutional Review Board
Commercial IRBs are a perfect example of a concourse career pathway that feels like a mystery to those on the outside. IRBs exist to protect the safety, rights, and welfare of trial participants. Entry-level opportunities vary, but many start on the operational side as an IRB analyst/specialist/coordinator, and strong regulatory and compliance backgrounds are often sought out as the foundation for a qualified IRB entry-level position.
Once employed in a commercial IRB, the opportunities for advancement range widely, each having unique requirements for education and experience levels. For example, experienced coordinators and investigators can become an IRB board member, but some boards may require specific medical expertise and relevant experience in conducting trials. Commercial IRBs provide a number of roles outside standard IRB responsibilities, including operations, business development, and project management.
Concourse G: Technology Solution Vendor
Technology solution vendors in the research industry represent a fast-growing career pathway. If you have attended any recent research conference, you will notice most new vendors in our space are driven by a range of technology-related challenges facing researchers.
The easiest way into this concourse is typically through project management, which would take some level of experience in the research industry. Once inside and you learn the specific technology solution or product, you can navigate toward more client-facing roles, such as adoption/implementation specialist, training, or sales. You can also pivot to more internal-focused positions, such as inside sales or operations.
Technology solution vendors are developing products and services that are designed to make conducting or managing research easier. So naturally, certain roles within these companies would benefit from specific research experience that may help those creating or designing their products to better understand the real-world implications.
Clinical Research Professional Personas
Now let’s look at some stories from the field. The personas below tell the story of how individuals may have entered their role and traversed the concourse.
 Landing in Pharma Olivia, Associate Director, Investigator Engagement Pharmaceutical Company |
Olivia studied biology at a university and worked in a lab focused on studying the impact of Alzheimer’s disease on mice. She earned a Master of Health Administration degree and landed a job at a large pediatric institution as a clinical research coordinator (CRC), where she worked her way up to a project manager position over the next seven years. She decided to move from her pediatric role into industry at a CRO, where she worked as a clinical research associate (CRA).Olivia later moved into the pharmaceutical industry, where she has worked for the past eight years in roles across clinical operations and global site management. Her focus is now on investigator engagement and strategic site partnerships to support successful clinical trial delivery. |
|
Mark didn’t know clinical research existed when he stumbled into his role as a CRC at a large pediatric hospital. He started to inquire with CRAs who monitored the site about how they got into their role. This opened his mind to the vast number of opportunities in the field. He started to explore roles in CROs that would allow him to “break in.”Although some of the CROs would only consider him for entry-level project coordinator, regulatory specialist, or CRA roles, he was able to find one that would hire him as a clinical trial manager, based on his education and experience. He accepted the position and started learning how to bridge the gap of knowledge between site and CRO operations in running clinical trials. |

Landing at a CRO |
|

Landing at a Commercial IRB |
Without even knowing what “IRB” stood for, Ben “fell into” the research industry by taking an administrative entry-level position at a large Commercial IRB. With a BA in Communications, he made copies of study protocols and informed consent documents, not fully understanding their contexts.After six months of making copies, Ben, through a networking connection at a large research university’s local IRB, took on an IRB coordinator role. His career flourished in positions focused on business development—he became Director of Clinical Research at a large AMC and later at a community research hospital, then an Associate Vice President for a research consulting firm, and finally a Vice President for a commercial IRB. |
|
 Landing at an AMC Sue, Senior Project Manager Academic Medical Center |
Sue earned her BS in Biochemistry and later earned a Master of Public Health degree. She worked for 11 years as a clinical research and data coordinator at a large AMC. She then moved into a project manager position for multisite clinical research and oversaw the management of a clinical research team and the clinical research portfolio for a pediatric hospital department.Sue now serves as Senior Project Manager of Operations in the Multisite Trials Coordinating Center at a large AMC. |
|
Conclusion
This concourse is a tool that provides awareness of the vast careers that clinical research professionals work in or may have a future interest in. Our goal in the creation of the tool and subsequent manuscript is to educate interested stakeholders and encourage outreach to showcase the ever-growing number of careers in clinical research. We want to narrow the gap from “falling into” this career to intentionally choosing clinical research as a future career path.
We admit that this airport concourse does not encompass all the major senior leadership roles that are out there, but it provides a moving walkway into the complexity of roles within clinical research. As another note, employer size does impact job responsibilities assigned to each employee, including new job opportunities within that employer.
Whether you feel like you are in a mad dash running through a concourse, catching the right flight at the right time, or a bit lost in the airport, it is important to remember there are always delays, easy direct flights, and concourse navigators ready to help you traverse your career.
Disclaimer
This publication was supported, in part, by The Ohio State University Clinical and Translational Science Institute (CTSI) and The University of Alabama at Birmingham Center for Clinical Translational Science (CCTS) under the National Center for Advancing Translational Sciences of the National Institutes of Health Grant Numbers UM1TR004548 and UM1TR004771. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
References
- Abbott A. 1988. The System of Professions: An Essay on the Division of Expert Labor. University of Chicago Press.
- Bailyn B. 1960. Education in the Forming of American Society. University of North Carolina Press.
- Susskind R, Susskind D. 2015. The Future of Professions: How Technology Will Transform the Work of Human Experts. Oxford University Press.
- Association of Clinical Research Professionals. 2025. Voices from the Front Lines: Insights from the Workforce on Transforming the Clinical Research Enterprise—Past, Present, and Future. https://acrpnet.org/insights/white-papers/voices-from-the-frontlines
- Freel SA, Snyder DC, Bastarache K, et al. 2023. Now is the time to fix the clinical research workforce crisis. Clinical Trials 20(5):457–62. doi:10.1177/17407745231177885
- Snyder DC, Gaudaur H, Marchant M, et al. 2024. Enhancing the clinical research workforce: a collaborative approach with human resources. Frontiers in Pharmacology doi:10.3389/fphar.2024.1295155
- Knapke JM, Snyder DC, Carter K, et al. 2022. Issues for recruitment and retention of clinical research professionals at academic medical centers: Part 1 – collaborative conversations Un-Meeting findings. Journal of Clinical and Translational Science 6(1):e80. https://pubmed.ncbi.nlm.nih.gov/35949656/; doi:10.1017/cts.2022.411
- Snyder DC, Brouwer RN, Ennis CL, et al. 2016. Retooling institutional support infrastructure for clinical research. Contemp Clin Trials 48:139-145. http://dx.doi.org/10.1016/j.cct.2016.04.010
- Sonstein SA, Seltzer J, Li R, Jones CT, Silva H, Daemen E. 2014. Moving from compliance to competency: A harmonized core competency framework for the clinical research professional. Clinical Researcher 28(3):17–23.
- Sonstein S, Brouwer RN, Gluck W, et al. 2020. Leveling the joint task force core competencies for clinical research professionals. Therapeutic Innovation and Regulatory Science 54(1):1–20. doi:10.1007/s43441-019-00024-2
- Fritter J, Jones CT. 2023. Mapping the pathway to take control of your clinical research career. Clinical Researcher 37(5). https://acrpnet.org/2023/10/17/mapping-the-pathway-to-take-control-of-your-clinical-research-career
- Association of Clinical Research Professionals. 2020. ACRP Career Lattice. https://acrpnet.org/wp-content/uploads/dlm_uploads/2020/08/Clincal-Research-Career-Lattice.pdf
- Fitzgerald D, Wilson C. 2024. What types of jobs and careers are there in the pharmaceutical and medical device industry? https://www.getreskilled.com/types-of-pharma-jobs/
- Waters L. 2024. 10 Clinical Research Career Paths. https://www.pharmiweb.jobs/article/10-clinical-research-career-paths
- ICON. 2024. A comprehensive guide to contract research organizations (CROs). https://careers.iconplc.com/blogs/2024-1/a-comprehensive-guide-to-contractresearch-organizations-cros
- Stanton K. 2022. Biotech vs Pharma: Differences and Similarities. https://www.qualio.com/blog/biotech-vs-pharma
- U.S. Food and Drug Administration Center for Devices and Radiological Health. 2022. How to determine if your product is a medical device. https://www.fda.gov/medical-devices/classify-your-medical-device/how-determine-if-your-product-medical-device
- Yeardley H. 2024. The CRA of the future: From ‘road warrior’ to site ‘owner.’ Applied Clinical Trials 33(1/2). https://www.appliedclinicaltrialsonline.com/view/the-cra-of-the-future-from-road-warrior-to-site-owner
Conflicts of Interest and Acknowledgement
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The authors further acknowledge the contributions of an anonymous reviewer toward the refinement of its contents.
Jessica Fritter, DHSc, MACPR, ACRP-CP, FACRP, (fritter.5@osu.edu) is an Associate Clinical Professor at the College of Nursing, Director of the Master of Clinical Research Program, Co-Director of Workforce Development, Clinical and Translational Science Institute, and Faculty Director, Multisite Trial Coordinating Center, The Ohio State University.
Susan P. Landis is Chief Executive Officer with the Association of Clinical Research Professionals.
Samantha Sharpe, MD, is a Senior Clinical Research Program Coordinator at Nationwide Children’s Hospital, and Clinical Assistant Professor of Practice at the College of Nursing, The Ohio State University.
Tia Patterson, PhD, is the Senior Learning Business Partner, Global Clinical Operations at BeOne Medicines, and Clinical Assistant Professor of Practice at the College of Nursing, The Ohio State University.
Kimberly McCall, PhD, is an Assistant Professor and Biotechnology Regulatory Affairs Specialist at the University of Alabama at Birmingham.
Samantha Martinek Bundt, MA, is the Clinical Research Professionals Workforce Development Project Manager for the Clinical and Translational Science Institute at The Ohio State University.
Tiffany Stoudemire, MCR, is Associate Director, Clinical Research Lead – Investigator Engagement, at Eli Lilly and Company
Justin Osborne, BA, (josborne@univo-group.com) is the Vice President of Consulting and Institutional Services at Univo IRB, a commercial IRB.
Carolynn T. Jones, DNP, MSPH, CRN-BC, FAAN, is a Professor and Coordinator, Clinical Research Management Certificate and MSN, The University of Alabama at Birmingham (UAB), School of Nursing, and Senior Scientist, UAB Center for Clinical Translational Science.



