Clinical Researcher—April 2026 (Volume 40, Issue 2)
RECRUITMENT & RETENTION
Alen Hadzic, MIE; Ryan Helber, MBA; James Prudden, BA
Clinical trial sites typically rely on patient populations culled from clinics, physician referrals, and reviews of electronic medical record files. These methods are effective to a point, but they are ineffective in identifying patients who are unknown to the healthcare system. Effective enrollment necessitates going beyond the purview of a medical system.
The need for diversity further complicates patient enrollment and adds financial stress that may hinder the success of a study. A targeted, engineered approach to patient enrollment and diversity, using a financially calculable strategy, can ameliorate these challenges.
Attaining Diversity: An Old Problem
There has been considerable research into the problems of attaining a diverse patient population.
Clinical trials need to reflect the true composition of a patient population. In 2015, for example, one-fifth of New Molecular Entities “were associated with differences in exposure, and/or response, across racial or ethnic groups, which resulted in prescribing recommendations for specific patient populations.”{1}
Black participants are the most consistently underrepresented subgroup in clinical trials, followed by Hispanic/Latino populations. White and Asian populations, meanwhile, are overrepresented.{1} Between 2015 and 2019, non-White patients accounted for less than 25% of all participants in clinical trials in the United States; only 13% of participants were Hispanic/Latino.{2}
The American Journal of Medicine observed that a study population “should reflect the epidemiology of the disease demographics and burden.” It further noted that race and ethnicity are social constructs and may best be understood through social determinants of health (SDOH), namely money, power, and resources. SDOH are affected by “education, income, employment, housing security, food security, transportation, social connection or isolation, physical activity, exposure to violence, immigration status, environmental and cultural influences, as well as healthcare….”{1}
Diversity increases the validity, safety, and applicability of clinical knowledge and determines whether findings generalize beyond a narrow subset of the population. Yet despite awareness, disparities persist. This recurring conundrum is not conceptual; however, it is operational.
A Global Phenomenon
Many clinical studies today are global in scope, but the problem of attaining a diverse patient population is global as well.{3} A study surveying respondents in 54 countries identified different concerns across populations. Asian respondents were more frequently concerned about time off from work and time required to participate. Hispanics were also more concerned about time off and expressed more concern about receiving placebo. Black and Hispanic respondents placed higher importance on diversity of staff members.{4}
A review of global cardiovascular trials underscored the economic implications of slow enrollment. These studies included at least 500 patients, had a median of 320 sites, and an enrollment period of 32 months, yet patient-per-site ratios remained low. The authors concluded, “In addition to slow enrollment, cardiovascular trials continue to have persistent underrepresentation of women, individuals in racial and ethnic minoritized groups, and rural patients.”{5}
Slow enrollment delays development timelines, prolongs operational costs, and increases financial risk. Diversity is therefore not solely an ethical concern. It is a measurable economic variable.
The Financial Architecture of Enrollment
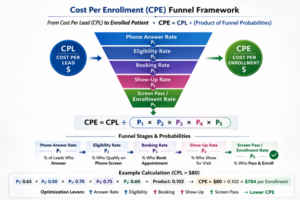
Sponsors do not purchase impressions; they purchase enrolled patients. The bridge between advertising expenditure and randomized participants is a probabilistic funnel.
Cost Per Lead (CPL) represents the cost required to generate one qualified expression of interest. Cost Per Enrollment (CPE) represents the cost required to produce one enrolled participant.
The correct formulation is:
CPE = CPL ÷ (product of funnel probabilities)
In clinical operations, these funnel probabilities correspond to measurable stages:
P₁ = Phone Answer Rate
P₂ = Eligibility Rate on Phone Screen
P₃ = Appointment Booking Rate
P₄ = Show-Up Rate
P₅ = Screen Pass/Enrollment Rate
The product of these five probabilities determines the proportion of leads that ultimately enroll. For example:
Phone Answer Rate = 0.65
Eligibility Rate = 0.50
Booking Rate = 0.70
Show-Up Rate = 0.75
Screen Pass/Enrollment Rate = 0.60
P Total = 0.65 × 0.50 × 0.70 × 0.75 × 0.60 = 0.102
If CPL is $80:
CPE = $80 ÷ 0.102
CPE = $784 per enrolled patient
The math reveals a critical insight: because CPE is divided by the product of probabilities, small inefficiencies compound dramatically.
If those same probabilities decline modestly:
0.55 × 0.40 × 0.60 × 0.65 × 0.50 = 0.0429
With the same $80 CPL:
CPE = $80 ÷ 0.0429
CPE = $1,865 per enrollment.
Advertising spend did not increase. Rather, funnel precision deteriorated. Diversity, therefore, is not inherently expensive. Misalignment between targeting and epidemiology is expensive.
Online Media Provide Highly Accurate Targeting Opportunities
Despite extensive research confirming lack of diversity and identifying limited actionable solutions, more sophisticated recruitment approaches have emerged. Digital platforms allow investigators to reach patients where they already spend time—online.
“Incorporating marketing techniques … can improve patient diversity, streamline recruitment processes, and ultimately help researchers meet their enrollment goals.”{6}
Patients research their condition online. Social media platforms analyze engagement patterns in real time. Using radial geo-targeting to assess proximity to research sites, combined with demographic overlays and epidemiological data from government sources, digital advertising becomes intentional rather than passive. Social media marketing campaigns are best handled by specialist, not generalist, agencies.{6}
“By combining disease state demographics, social media platform user data, and regional demographic information, you can predict and achieve a representative participant pool.”{6}
Social media algorithms provide highly precise targeting. If you know the prevalence of a certain population lies mostly within a specific demographic, and know the demographics of a particular city, then digital advertising can prioritize these characteristics.
When targeting aligns with disease prevalence:
- Phone answer rates increase because messaging resonates.
• Eligibility rates improve because outreach goes to higher prevalence groups.
• Booking rates rise because expectations are clear.
• Show-up rates improve when logistical barriers are anticipated.
• Screen pass/enrollment rates stabilize because pre-qualification is accurate.
The product of funnel probabilities increases.
Because CPE = CPL ÷ (product of funnel probabilities), improvements in these downstream variables directly reduce enrollment costs (see figure below).
Diversity as an Economically Calculable Variable
The enrollment cost of diversity is not a theoretical construct; it is the output of a measurable equation.
If outreach fails to reach underrepresented communities, eligibility rates decline and screen failures increase. If trust barriers are not addressed, answer rates fall. If transportation constraints are ignored, show-up rates decrease. Each decline reduces the denominator in the CPE equation and inflates cost per enrollment.
Conversely, when recruitment is engineered with demographic precision, funnel performance stabilizes. Cost becomes forecastable, budget allocation becomes rational, and accrual timelines become predictable. Diversity transitions from aspiration to infrastructure.
Conclusion
The effort to increase diversity in clinical trial enrollment can be engineered with a highly targeted digital marketing campaign that uses the capabilities of social media to identify with high accuracy the most appropriate candidates.
When recruitment is translated into CPL and divided by measurable probabilities—phone answer rate, eligibility rate, booking rate, show-up rate, and screen pass/enrollment rate—cost per enrollment becomes quantifiable.
The enrollment cost of diversity is therefore not an unpredictable premium. It is the measurable output of funnel efficiency.
When the funnel is engineered to reflect true epidemiology, diversity becomes financially modellable, operationally controllable, and statistically aligned with the populations most affected by disease.
Credits
The authors would like to thank the following organizations: Advanced Pharma Clinical Research, Miami, Fla.; and Rogans Salud Site Network, Bogota, Colombia.
References
- Rodney RA. 2021. The Attainment of Patient Diversity in Clinical Trials: Race, Ethnicity, Genetics. Am J Med 134(12):1440–1. https://www.amjmed.com/article/S0002-9343(21)00488-5/fulltext
- Chapman-Davis E, Webster EM, Hines JF. 2023. Achieving diversity in clinical trial enrollment by reducing burden and increasing value: A patient-centered approach. Gynecol Oncol 172:A1–2. https://www.gynecologiconcology-online.net/article/S0090-8258(23)00191-9/abstract
- Khan MH, Ibanez KR, Bowen C, et al. 2025. Impact of Global Enrollment on Race, Ethnicity, and Age Representation in Pivotal Gynecologic Cancer Trials Leading to US Food and Drug Administration Drug Approvals. JCO Glob Oncol 11:e2400636. doi:10.1200/GO-24-00636
- de Bruin A, Masullo J, Sine S, et al. 2025. Promoting Diversity through an Understanding of Barriers and Drivers for Inclusive Clinical Trials. Ther Innov Regul Sci 59(3):596–605. doi:10.1007/s43441-025-00751-9
- Khan MS, Jamil A, Shakoor M. 2025. Patient Enrollment for Cardiovascular Clinical Trials in the United States. JAMA Cardiol 10(3):298–300. doi:10.1001/jamacardio.2024.5537
- Hadzic A, Brathwaite JS, Sheth M, et al. 2024. Social Media Marketing and its Vital Role in Improving Clinical Trial Recruitment. Clin Researcher 38(6). https://acrpnet.org/2024/12/13/social-media-marketing-and-its-vital-role-in-improving-clinical-trial-recruitment

Alen Hadzic, MIE, is CEO of CT Scan.
Ryan Helber, MBA, is Chief Strategy Officer of CT Scan.
James Prudden, BA, is Director of Communications of CT Scan.



